|
The Third Chimpanzee
In PAOLA CAVALIERI & PETER SINGER (eds.), The Great Ape Project
New York: St. Martin's Griffin, 1993, pp. 89-101
The next time
that you visit a zoo, make a point of walking past the ape cages.
Imagine that the apes had lost most of their hair, and imagine a
cage nearby holding some unfortunate people who had no clothes and
couldn't speak but were otherwise normal. Now try guessing how
similar those apes are to ourselves genetically. For instance,
would you guess that a chimpanzee shares 10, 50 or 99 per cent of
its genes with humans?
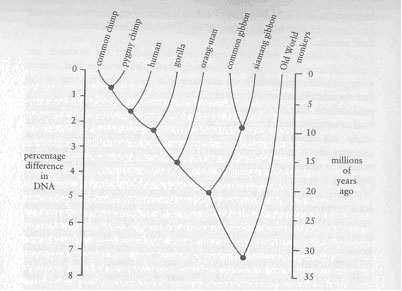
Then ask yourself why those apes are on exhibition in cages, and why other apes are being used for medical experiments, while it is not permissible to do either of those things to humans. Suppose it turned out that chimps shared 99.9 per cent of their genes with us, and that the important differences between humans and chimps were due to just a few genes. Would you still think it is okay to put chimps in cages and to experiment on them? Consider those unfortunate mentally impaired people who have much less capacity to solve problems, to care for themselves, to communicate, to engage in social relationships and to feel pain, than do apes. What is the logic that forbids medical experiments on those people, but not on apes? You might answer that apes are 'animals', while humans are humans, and that is enough. An ethical code for treating humans should not be extended to an 'animal', no matter what percentage of its genes it shares with us, and no matter what its capacity for social relationships or for feeling pain. That is an arbitrary but at least self-consistent answer that cannot be lightly dismissed. In that case, learning more about our ancestral relationships will not have any ethical consequences, but it will satisfy our intellectual curiosity to understand where we come from. Every human society has felt a deep need to make sense of its origins, and has answered that need with its own story of the Creation. What follows is the creation story of our time. For centuries it has been clear approximately where we fit into the animal kingdom. We are obviously mammals, the group of animals characterised by having hair, nursing their young and other features. Among mammals we are obviously primates, the group of mammals including monkeys and apes. We share with other primates numerous traits lacking in most other mammals, such as flat fingernails and toenails rather than claws, hands for gripping, a thumb that can be opposed to the other four fingers and a penis that hangs free rather than being attached to the abdomen. By the second century AD, the Greek physician Galen had deduced our approximate place in nature correctly when he dissected various animals and found that a monkey was 'most similar to man in viscera, muscles, arteries, veins, nerves and in the form of bones'. It is also easy to place us within the primates, among which we are obviously more similar to apes than to monkeys. To name only one of the most visible signs, monkeys sport tails, which, like the apes, we lack. It is also clear that gibbons, with their small size and very long arms, are the most distinctive apes, and that orang-utans, chimpanzees, gorillas and humans are all more closely related to each other than any of them is to gibbons. But to go further with our relationships proves unexpectedly difficult. It has provoked an intense scientific debate, which revolves around three questions including the one that I posed in the first paragraph in this chapter: 1. What is the detailed family tree of relationships among humans, the living apes and extinct ancestral apes? For example, which of the living apes is our closest relative? 2. When did we and that closest living relative, whichever ape it is, diverge from a common ancestor? 3. What fraction of our genes do we share with that closest living relative? At first, it would seem natural to assume that comparative anatomy had already solved the first of those three questions. We look especially like chimpanzees and gorillas, but differ from them in obvious features such as our larger brains, upright posture and much sparser body hair as well as in many more subtle points. However, on closer examination these anatomical facts are not decisive. Depending on what anatomical characters one considers most important and how one interprets them biologists differ on whether we are most closely related to the orangutan (the minority view), with chimps and gorillas having branched off our family tree before we split off from orang-utans, or whether we are instead closest to chimps and gorillas (the majority view), with the ancestors of orang-utans having gone their separate way earlier. Within the majority, most biologists have thought that gorillas and chimps are more like each other than either is like us, implying that we branched off before the gorillas and chimps diverged from each other. This conclusion reflects the common-sense view that chimps and gorillas can be lumped in a category termed 'apes', while we are something different. However, it is also conceivable that we look distinct only because chimps and gorillas have not changed much since we diverged from our common ancestor, while we have changed greatly in a few important and highly visible features, such as upright posture and brain size. In that case, humans might be most similar to gorillas, or humans might be most similar to chimps, or humans and gorillas and chimps might be roughly equidistant from each other, in overall genetic make-up. Hence, anatomists have continued to argue about the first question, the details of our family tree. Whichever tree one prefers, anatomical studies by themselves tell us nothing about the second and third questions, our time of divergence and genetic distance from apes. Perhaps fossil evidence might in principle solve the questions of the correct ancestral tree and of dating, though not the question of genetic distance. If we had abundant fossils, we might hope to find a series of dated proto-human fossils and another series of dated proto-chimp fossils converging on a common ancestor around ten million years ago, converging in turn on a series of proto-gorilla fossils twelve million years ago. Unfortunately, that hope for insight from the fossil record has also been frustrated, because almost no ape fossils of any sort have been found for the crucially relevant period between five and fourteen million years ago in Africa. The solution to these questions about our origins came from an unexpected direction: molecular biology as applied to bird taxonomy. About thirty years ago, molecular biologists began to realise that the chemicals of which plants and animals are composed might provide 'clocks' by which to measure genetic distances and to date times of evolutionary divergence. The idea is as follows. Suppose there is some class of molecules that occurs in all species, and whose particular structure in each species is genetically determined. Suppose further that structure changes slowly over the course of millions of years because of genetic mutations, and that the rate of change is the same in all species. Two species derived from a common ancestor would start off with identical forms of the molecule, which they inherited from that ancestor, but mutations would then occur independently and produce structural changes between the molecules of the two species. The two species' versions of the molecule would gradually diverge in structure. If we knew how many structural changes now occur on the average every million years, we could then use the difference today in the molecule's structure between any two related animal species as a clock, to calculate how much time had passed since the species shared a common ancestor. For instance, suppose one knew from fossil evidence that lions and tigers diverged five million years ago. Suppose the molecule in lions were 99 per cent identical in structure to the corresponding molecule in tigers and differed only by 1 per cent. If one then took a pair of species of unknown fossil history and found that the molecule differed by 3 per cent between those two species, the molecular clock would say that they had diverged three times five million, or fifteen million, years ago. Neat as this scheme sounds on paper, testing whether it succeeds in practice has cost biologists much effort. Four things had to be done before molecular clocks could be applied: find the best molecule; find a quick way of measuring changes in its structure; prove that the clock runs steady (that is, that the molecule's structure really does evolve at the same rate among all the species that one is studying); and measure what that rate is. Molecular biologists had worked out the first two of these problems by around 1970. The best molecule proved to be deoxyribonucleic acid (abbreviated to DNA), the famous substance whose structure James Watson and Francis Crick showed to consist of a double helix, thereby revolutionising the study of genetics. DNA is made up of two complementary and extremely long chains, each made up of four types of small molecules whose sequence within the chain carries all the genetic information transmitted from parents to offspring. A quick method of measuring changes in DNA structure is to mix the DNA from two species, then to measure by how many degrees of temperature the melting-point of the mixed (hybrid) DNA is reduced below the melting-point of pure DNA from a single species. Hence the method is generally referred to as DNA hybridisation. As it turns out, a melting-point lowered by one degree Celsius (abbreviated: delta T = 1°C) means that the DNAs of the two species differ by roughly 1 per cent. In the 1970s, most molecular biologists and most taxonomists had little interest in each other's work. Among the few taxonomists who appreciated the potential power of the new DNA hybridisation technique was Charles Sibley, an ornithologist then serving as Professor of Ornithology and Director at Yale's Peabody Museum of Natural History. Bird taxonomy is a difficult field because of the severe anatomical constraints imposed by flight. There are only so many ways to design a bird capable, say, of catching insects in mid-air, with the result that birds of similar habits tend to have very similar anatomies, whatever their ancestry. For example, American vultures look and behave much like Old World vultures, but biologists have come to realise that the former are related to storks, the latter to hawks, and that their resemblances result from their common lifestyle. Frustrated by the shortcomings of traditional methods for deciphering bird relationships, Sibley and Jon Ahlquist turned in 1973 to the DNA clock, in the most massive application to date of the methods of molecular biology to taxonomy. Not until 1980 were Sibley and Ahlquist ready to begin publishing their results, which eventually came to encompass applying the DNA clock to about 1,700 bird species - nearly one-fifth of all living birds. While Sibley's and Ahlquist's achievement was a monumental one, it initially caused much controversy because so few other scientists possessed the blend of expertise required to understand it. Here are typical reactions I heard from my scientist friends: 'I'm sick of hearing about that stuff. I no longer pay attention to anything those guys write' (an anatomist). 'Their methods are OK, but why would anyone want to do something so boring as all that bird taxonomy?' (a molecular biologist). 'Interesting, but their conclusions need a lot of testing by other methods before we can believe them' (an evolutionary biologist). 'Their results are The Revealed Truth, and you'd better believe it' (a geneticist). My own assessment is that the last view will prove to be the most nearly correct one. The principles on which the DNA clock rests are unassailable; the methods used by Sibley and Ahlquist are state-of-the-art; and the internal consistency of their genetic-distance measurements from over 18,000 hybrid pairs of bird DNA testifies to the validity of their results. Just as Darwin had the good sense to marshal his evidence for variation in barnacles before discussing the explosive subject of human variation, Sibley and Ahlquist similarly stuck to birds for most of the first decade of their work with the DNA clock. Not until 1984 did they publish their first conclusions from applying the same DNA methods to human origins, and they refined their conclusions in later papers. Their study was based on DNA from humans and from all of our closest relatives: the common chimpanzee, pygmy chimpanzee, gorilla, orangutan, two species of gibbons and seven species of Old World monkeys. Figure 8.1 summarises the results. As any anatomist would have predicted, the biggest genetic difference, expressed in a big DNA melting-point lowering, is between monkey DNA and the DNA of humans or of any ape. This simply puts a number on what everybody has agreed ever since apes first became known to science: that humans and apes are more closely related to each other than either are to monkeys. The actual statistic is that monkeys share 93 per cent of their DNA structure with humans and apes, and differ in 7 per cent. Equally unsurprising is the next biggest difference, one of 5 per cent between gibbon DNA and the DNA of other apes or humans. This too confirms the accepted view that gibbons are the most distinct apes, and that our affinities are instead with gorillas, chimpanzees and orangutans. Among those latter three groups of apes, most recent anatomists have considered the orang-utan as somewhat separate, and that conclusion too fits the DNA evidence: a difference of 3.6 per cent between orang-utan DNA and that of humans, gorillas and chimpanzees. Geography confirms that the latter three species parted from gibbons and orang-utans quite some time ago: living and fossil gibbons and orangutans are confined to Southeast Asia, while living gorillas and chimpanzees plus early fossil humans are confined to Africa. At the opposite extreme but equally unsurprising, the most similar DNAs are those of common chimpanzees and pygmy chimpanzees, which are 99.3 per cent identical and differ by only 0.7 per cent. So similar are these two chimp species in appearance that it was not until L929 that anatomists even bothered to give them separate names. Chimps living on the equator in central Zaire rate the name 'pygmy chimps' because they are on average slightly smaller (and have more slender builds and longer legs) than the widespread 'common chimps' ranging across Africa just north of the equator. However, with the increased knowledge of chimp behaviour acquired in recent years, it has 'come clear that the modest anatomical differences between pygmy and common chimps mask considerable differences in reproductive biology. Unlike common chimps, but like ourselves, pygmy chimps assume a wide variety of positions for copulation, including face-to-face; copulation can be initiated by either sex, not just by the male; females are sexually receptive for much of the month, not just for a briefer period in mid-month; and there are strong bonds among females or between males and females, not just among males. Evidently, those few genes (0.7 per cent) that differ between pygmy and comrron chimps have big consequences for sexual physiology and roles. That same theme — a small percentage of gene differences having great consequences — will recur later in this chapter with regard to the gene differences between humans and chimps.
Figure 8.1 Trace back each pair of modern higher primates to the black dot connecting them. The numbers to the left then give the percentage difference between the DNAs of those modern primates, while the numbers to the right give the estimated number of millions of years since they diverged from a common ancestor. For example, the common and pygmy chimps differ in about 0.7% of their DNA and diverged about three million years ago; we differ in about 1.6% of our DNA from both species of chimps and diverged from their common ancestor about seven million years ago; gorillas differ in about 2.3% of their DNA from us and from chimps and diverged from the common ancestor leading to us and the two chimps about ten million years ago. In all cases that I have discussed so far, anatomical evidence of relationships was already convincing, and the DNA-based conclusions confirmed what the anatomists had already concluded. But DNA was also able to resolve the problem at which anatomy had failed – the relationships between humans, gorillas and chimpanzees. As Figure 8.1 shows humans differ from both common chimps and pygmy chimps in about 1.6 per cent of their (our) DNA, and share 98.4 per cent. Gorillas differ somewhat more, by about 2.3 per cent, from us and from both of the chimps. Let us pause to let some of the implications of these momentous numbers sink in. The gorilla must have branched off from our family tree slightly before we separated from the common and pygmy chimpanzees. The chimpanzees, not the gorilla, are our closest relatives. Put another way, the chimpanzees' closest relative is not the gorilla but the human. Traditional taxonomy has reinforced our anthropocentric tendencies by claiming to see a fundamental dichotomy between mighty man, standing alone on high, and the lowly apes all together in the abyss of bestiality. Now future taxonomists may see things from the chimpanzees' perspective: a weak dichotomy between slightly higher apes (the three chimpanzees, including the 'human chimpanzee') and slightly lower apes (gorilla, orang-utan, gibbon). The traditional distinction between 'apes' (defined as chimps, gorillas, etc.) and humans misrepresents the facts. The genetic distance (1.6 per cent) separating us from pygmy or common chimps is barely double that separating pygmy from common chimps (0.7 per cent). It is less than that between two species of gibbons (2.2 per cent), or between such closely related North American bird species as red-eyed vireos and white-eyed vireos (2.9 per cent), or between such closely related and hard-to-distinguish European bird species as willow warblers and chiffchaffs (2.6 per cent). The remaining '8.4 per cent of our genes are just normal chimp genes. For example, our principal haemoglobin, the oxygen-carrying protein that gives blood its red colour, is identical in all 287 units with chimp haemoglobin. In this respect as in most others, we are just a third species of chimpanzee, and what is good enough for common and pygmy chimps is good enough for us. Our important visible distinctions from the other chimps – our upright posture, large brains, ability to speak, sparse body air, and peculiar sexual lives - must be concentrated in a mere 1.6 per cent of our genes. If genetic distances between species accumulated at a uniform rate ith time, they would function as a smoothly ticking clock. All that would be required to convert genetic distance into absolute time since the last common ancestor would be a calibration, furnished by a pair of species for which we know both the genetic distance and the time of divergence as dated independently by fossils. In fact, two independent calibrations are available for higher primates. On the one hand, monkeys diverged from apes between twenty-five and thirty million years ago, according to fossil evidence, and now differ in about 7.3 per cent of their DNA. On the other hand, orang-utans diverged from chimps and gorillas between twelve and sixteen million years ago and now differ in about 3.6 per cent of their DNA. Comparing these two examples, a doubling of evolutionary time, as one goes from twelve or sixteen to twenty-five or thirty million years, leads to a doubling of genetic distance (from 3.6 to 7.3 per cent of DNA). Thus, the DNA clock has ticked relatively steadily among higher primates. With those calibrations, Sibley and Ahlquist estimated the following time scale for our evolution. Since our own genetic distance from chimps (1.6 per cent) is about half the distance of orang-utans from chimps (3.6 per cent), we must have been going our separate way for about half of the twelve to sixteen million years that orang-utans had to accumulate their genetic distinction from chimps. That is, the human and 'other chimp' evolutionary lines diverged around six to eight million years ago. By the same reasoning, gorillas parted from the common ancestor of us three chimpanzees around nine million years ago, and the pygmy and common chimps diverged around three million years ago. In contrast, when I took physical anthropology as a college freshman in 1954, the assigned textbooks said that humans diverged from apes fifteen to thirty million years ago. Thus, the DNA clock strongly supports a controversial conclusion also drawn from several other molecular clocks based on amino acid sequences of proteins, mitochondria! DNA and globin pseudogene DNA. Each clock indicates that humans have had only a short history as a species distinct from other apes, much shorter than paleontologists used to assume. What do these results imply about our position in the animal kingdom? Biologists classify living things in hierarchical categories, each less distinct than the next: subspecies, species, genus, family, superfamily, order, class and phylum. The Encyclopaedia Britannica and all the biology texts on my shelf say that humans and apes belong to the same order, called Primates, and the same superfamily, called Hominoidea, but to separate families, called Hominidae and Pongidae. Whether Sibley's and Ahlquist's work changes this classification depends on one's philosophy of taxonomy. Traditional taxonomists group species into higher categories by making somewhat subjective evaluations of how important the differences between species are. Such taxonomists lace humans in a separate family because of distinctive functional traits place large brain and bipedal posture and this classification would remain unaffected by measures of genetic distance. However, another school of taxonomy, called cladistics, argues that classification should be objective and uniform, based on genetic distance or times of divergence. All taxonomists agree now that red-eyed and white-eyed vireos belong together in the genus Vireo, willow warblers and chiffchaffs in the genus Phylloscopus, the various species of gibbons in the genus Hylobates. Yet the members of each of these pairs of species are genetically more distant from each other than are humans from the other two chimpanzees, and diverged longer ago. On this basis, then, humans do not constitute a distinct family, nor even a distinct genus, but belong in the same genus as common and pygmy chimps. Since our genus name Homo was proposed first, it takes priority, by the rules of zoological nomenclature, over the genus name Pan coined for the 'other' chimps. Thus, there are not one but three species of genus Homo on Earth today: the common chimpanzee, Homo troglodytes; the pygmy chimpanzee, Homo paniscus; and the third chimpanzee or human chimpanzee, Homo sapiens. Since the gorilla is only slightly more distinct, it has almost equal right to be considered a fourth species of Homo. Even taxonomists espousing cladistics are anthropocentric, and the lumping of humans and chimps into the same genus will undoubtedly be a bitter pill for them to swallow. There is no doubt, however, that whenever chimpanzees learn cladistics, or whenever taxonomists from outer space visit Earth to inventory its inhabitants, they will unhesitatingly adopt the new classification. Which particular genes are the ones that differ between humans and chimps? Before we can consider this question, we need first to understand what it is that DNA, our genetic material, does. Much or most of our DNA has no function and may just constitute 'molecular junk': that is, DNA molecules that have become duplicated or have lost former functions, and that natural selection has not eliminated from us because they do us no harm. Of our DNA that does have known functions, the main ones have to do with the long chains of amino acids called proteins. Certain proteins make up much of our body's structure (such as the proteins keratin, of hair, or collagen, of connective tissue), while other proteins, termed enzymes, synthesise and break down most of our body's remaining molecules. The sequences of the component small molecules (nucleotide bases) in DNA specify the sequence of amino acids in our proteins. Other parts of our functional DNA regulate protein synthesis. Those of our observable features that are easiest to understand genetically are ones arising from single proteins and single genes. For instance, our blood's oxygen-carrying protein haemoglobin, already mentioned, consists of two amino acid chains, each specified by a single chunk of DNA (a single 'gene'). These two genes have no observable effects except through specifying the structure of haemoglobin, which is continued to our red blood cells. Conversely, haemoglobin's structure is totally specified by those genes. What you eat or how much you exercise may affect how much haemoglobin you make, but not the details of its structure. That is the simplest situation, but there are also genes influencing many observable traits. For example, the fatal genetic disorder known as Tay-Sachs disease involves many behavioural as well as anatomical anomalies: excessive drooling, rigid posture, yellowish skin, abnormal head growth and other symptoms. We know in this case that all these observable effects result somehow from changes in a single enzyme specified by the Tay-Sachs gene, but we do not know exactly how. Since that enzyme occurs in many tissues of our bodies and breaks down a widespread cellular constituent, changes in that one enzyme have wide-ranging and ultimately fatal consequences. Conversely, some traits, such as your height as an adult, are influenced simultaneously by many genes and also by environmental factors (for example, your nutrition as a child). While scientists understand well the function of numerous genes that specify known individual proteins, we know much less about the function of genes involved in more complex determinations of traits, such as most behavioural features. It would be absurd to think that human hallmarks such as art, language or aggression depend on a single gene. Behavioural differences among individual humans are obviously subject to enormous environmental influences, and what role genes play in such individual differences is a controversial question. However, for those consistent behavioural differences between chimps and humans, genetic differences are likely to be involved in those species' differences, even though we cannot yet specify the genes responsible. For instance, the ability of humans but not chimps to speak surely depends on differences in genes specifying the anatomy of the voice box and the wiring of the brain. A young chimpanzee brought up in a psychologist's home along with the psychologist's human baby of the same age still continued to look like a chimp and did not learn to talk or walk erect. But whether an individual human grows up to be fluent in English or Korean is independent of genes and dependent solely on its childhood linguistic environment, as proved by the linguistic attainments of Korean infants adopted by English-speaking parents. With this as background, what can we say about the 1.6 per cent of our DNA that differs from chimp DNA? We know that the genes for our principal haemoglobin do not differ, and that certain other genes do exhibit minor differences. In the nine protein chains studied to date in both humans and common chimps, only five out of a total of 1,271 amino acids differ: one amino acid in a muscle protein called myoglobin one in a minor haemoglobin chain called the delta chain and three in an enzyme called carbonic anhydrase. But we do not yet know which chunks of our DNA are responsible for the functionally significant differences between humans and chimps, such as differences in brain size anatomy of the pelvis, voice box and genitalia, amount of body hair, female menstrual cycle, menopause and other traits. Those important differences certainly do not arise from the five amino acid differences detected to date. At present, all we can say with confidence is this: much of our DNA is junk; at least some of the 1.6 per cent that differs between us and chimps is already known to be junk; and the functionally significant differences must be confined to some as yet unidentified small fraction of 1.6 per cent. While we do not know which particular genes are the crucial ones, there are numerous precedents for one or a few genes having a big impact. I just mentioned the many large and visible differences between Tay-Sachs patients and other people, all somehow arising from a single change in one enzyme. That is an example of differences among individuals of the same species. As for differences between related species, a good example is provided by the cichlid fishes of Africa's Lake Victoria. Cichlids are popular aquarium species, of which about 200 are confined to that one lake, where they evolved from a single ancestor within perhaps the last 200,000 years. Those 200 species differ among themselves in their food habits as much as do tigers and cows. Some graze on algae, others catch other fish, and still others variously crush snails, feed on plankton, catch insects, nibble the scales off other fish or specialise in grabbing fish embryos from brooding mother fish. Yet all those Lake Victoria cichlids differ from each other on the average by only about 0.4 per cent of their DNA studied. Thus it took even fewer genetic mutations to change a snail-crusher into a specialised baby-killer than it took to produce us from an ape. Do the new results about our genetic distance from chimps have any broader implications, besides technical questions of taxonomic names? Probably the most important implications concern how we think about the place of humans and apes in the universe. Names are not just technical details but express and create attitudes. The new results do not specify how we should think about humans and apes, but, just as did Darwin's On the Origin of Species, they will probably influence how we do think, and it will probably take us many years to readjust our attitudes. At present we make a fundamental distinction between animals (including apes) and humans, and this distinction guides our ethical code and actions. For instance, as I noted at the start of this chapter, it is considered acceptable to exhibit caged apes in zoos, but it is not acceptable to do the same with humans. I wonder how the public will feel when the identifying label on the chimp cage in the zoo reads ''Homo troglodytes'. I also noted earlier that it is considered acceptable to subject apes, but not humans, without their consent to lethal experiments for purposes of medical research. The motive for doing so is precisely because apes are so similar to us genetically. They can be infected with many of the same diseases as we can, and their bodies respond similarly to the disease organisms. Thus, experiments on apes offer a far better way to devise improved medical treatment for humans than would experiments on any other animals. There is no socially accepted human analogue of medical research on animals, even though lethal experiments on humans would provide medical scientists with far more valuable information than do lethal experiments on chimps. Yet the human experiments performed by Nazi concentration camp physicians are widely viewed as one of the most abominable of all the Nazis' abominations. Why is it all right to perform such experiments on chimps? Somewhere along the scale from bacteria to humans, we have to decide where killing becomes murder, and where eating becomes cannibalism. Most people draw those lines between humans and all other species. However, many people are vegetarians, unwilling to eat any animal (yet willing to eat plants). An increasingly vocal minority, belonging to the animal rights movement, objects to medical experiments on animals - or at least on certain animals. That movement is especially indignant at research on cats and dogs and primates, but generally silent about insects and bacteria. If our ethical code makes a purely arbitrary distinction between humans and all other species, then we have a code based on naked selfishness devoid of any higher principle. If our code instead makes distinctions based on our superior intelligence, social relationships and capacity for feeling pain, then it becomes difficult to defend an all-or-nothing code that draws a line between all humans and all animals. Instead, different ethical constraints should apply to research on different species. Perhaps it is just our naked selfishness, re-emerging in a new that would advocate granting special rights to those animal frenetically closest to us. But an objective case, based on the considerations I have just mentioned (intelligence, social relationships, etc.) can be made that chimps and gorillas qualify for preferred ethical consideration over insects and bacteria. If there is any animal species currently used in medical research for which a total ban on medical experimentation can be justified, that species is surely the chimpanzee. |
||||||||||||